What Is a Proprietary Blend? The Legal Meaning and Why It Matters

Last updated: April 20, 2026
A proprietary blend is a combination of multiple ingredients listed on a supplement or product label as a single combined weight, without disclosing the individual amount of each ingredient. The Dietary Supplement Health and Education Act of 1994 (DSHEA) permits this labeling format, which means consumers cannot tell from the label how much of each component the product actually contains. Proprietary blends are most common in pre-workout supplements, fat burners, testosterone boosters, and nootropics.
What is a proprietary blend?
On a dietary supplement's Supplement Facts panel, a proprietary blend appears as a named combination — for example, "TrueActive Performance Blend: 1,200 mg" — followed by a list of ingredients in descending order by weight. The total weight is disclosed. The individual weights of each listed ingredient are not.
This format is legal under the Dietary Supplement Health and Education Act of 1994 (DSHEA) and FDA regulations at 21 CFR 101.36. The regulation allows supplement manufacturers to protect what they describe as trade secrets by not disclosing the exact proportion of each ingredient in a blend.
The name "proprietary blend" signals that the specific recipe is proprietary — owned by the manufacturer — and therefore concealed. In practice, the concealment is not about protecting genuine formulation secrets. Most proprietary blends use well-known ingredients at undisclosed doses. The "proprietary" framing is primarily a marketing signal suggesting exclusivity and a regulatory cover that permits under-dosing.
Why do brands use proprietary blends?
The business case for proprietary blends comes down to three factors:
1. Cost control. Effective doses of many functional ingredients are expensive. Caffeine, creatine, and beta-alanine at clinically studied doses raise the cost of goods significantly. A proprietary blend lets a brand include a trace amount of an expensive ingredient — enough to list it on the label — while using higher doses of cheaper filler ingredients to make up the total blend weight.
2. Label concealment. When individual doses are disclosed, consumers and competitors can compare products directly. A pre-workout that lists 200 mg of caffeine competes on that number against every other pre-workout. Hidden in a proprietary blend, the same 50 mg of caffeine becomes invisible and beyond comparison.
3. Perceived exclusivity. "Proprietary" suggests that the formula is distinctive, tested, and worth its price. Consumers pay a premium for branded blends even when the underlying ingredients are commodity.
A fourth, narrower reason exists: brands occasionally claim that undisclosed ratios protect genuine trade secrets. This is rare. Genuine trade secrets in supplement formulation are unusual because ingredient interactions are well-documented in peer-reviewed literature.
Is "proprietary blend" a legally protected term?
The term "proprietary blend" is not itself legally defined. What is defined is the format under which such blends can be disclosed on a Supplement Facts panel. FDA regulation 21 CFR 101.36(c) requires that:
- Ingredients be listed in descending order by weight
- The total weight of the blend be disclosed
- Each ingredient in the blend be listed by its common or usual name
Nothing in the regulation requires the individual ingredient weights to be disclosed. This is the legal basis for the entire proprietary blend concealment practice.
Outside of dietary supplements, the term "proprietary" has even less legal specificity. A skincare product, a household cleaner, or an appliance can be labeled with a "proprietary formula" or "proprietary technology" with no regulatory substantiation required — though any claim about what the proprietary formulation does is subject to FTC prior substantiation rules.
What are the rules on labeling proprietary blends?
For dietary supplements specifically:
- Required disclosures: All ingredients in the blend must be listed by name in descending order of weight. The total blend weight must be disclosed. Any ingredient that appears separately on the label (not part of the blend) must have its individual weight disclosed per standard Supplement Facts rules.
- Prohibited practices: A brand cannot include an ingredient in a blend without listing it. Any ingredient that is present in amounts large enough to have a pharmacological effect typically must be disclosed under a combination of FDA rules and FTC substantiation requirements.
- Testing obligations: Third-party certification programs like NSF International, USP Dietary Supplement Verification, and Informed Sport test for label accuracy. Brands that participate in these programs submit their products for verification that what's on the label matches what's in the product. Brands without third-party certification are not subject to this independent verification.
- Good Manufacturing Practices (GMP): FDA's GMP regulations for dietary supplements (21 CFR Part 111) require manufacturers to establish specifications for the identity, purity, strength, and composition of their products. This applies to proprietary blends as well — the manufacturer internally must know what's in the blend even if consumers don't.
How can consumers see what's actually in a proprietary blend?
In most cases, full disclosure requires manufacturer cooperation or independent lab testing. Practical options:
1. Check for third-party testing certifications. NSF Certified for Sport, USP Verified, and Informed Sport seals indicate that the product has been tested for label accuracy and contaminants. These programs typically do not publish the full ingredient amounts, but they confirm that what the manufacturer states is accurate.
2. Estimate from the order of ingredients. Ingredients are listed by descending weight. If a proprietary blend is 500 mg total and lists six ingredients, the first ingredient must be at least 1/6 of the total (83 mg) and the last ingredient may be less than 10 mg. This does not give precise amounts but provides floors and ceilings.
3. Look for published formulation disclosures. Some brands publish full formulations voluntarily, often on their product page or a "transparency" page. Brands that do this are signaling confidence in their doses; brands that don't are signaling the opposite.
4. Check Labdoor and ConsumerLab. Both organizations independently test supplements and publish results, sometimes including component identification. Coverage is not universal but is growing.
5. Compare to research-verified doses. If a blend claims to contain caffeine and the full blend weight is 200 mg across 12 ingredients, the caffeine dose is almost certainly below the 100-200 mg range most caffeine research uses. This kind of reasoning can reveal under-dosing even without exact amounts.
Recent enforcement actions on proprietary blend misuse
The FTC and FDA rarely bring cases specifically targeting the proprietary blend format itself, because the format is explicitly permitted by DSHEA. Enforcement typically focuses on associated claims — when a brand claims a proprietary blend produces a specific effect, and the underlying formulation cannot plausibly support that effect.
Class actions have been brought against supplement brands on theories including: fraudulent concealment of under-dosed ingredients, deceptive labeling in states with stricter consumer protection laws than DSHEA (notably California and New York), and breach of warranty when blend compositions diverged materially from reasonable consumer expectations.
This section is updated as new relevant enforcement actions are documented.
Frequently asked questions
What does proprietary blend mean? A proprietary blend is a combination of multiple ingredients listed as a single combined weight on a supplement label, without disclosing individual ingredient amounts. Permitted under DSHEA (21 CFR 101.36).
Why do supplement brands use proprietary blends? To reduce cost by under-dosing expensive ingredients, to avoid direct comparison with competitors, and to signal exclusivity.
Are proprietary blends safe? Not inherently unsafe, but the transparency gap makes safety assessment harder — particularly for consumers taking medications that may interact with individual components.
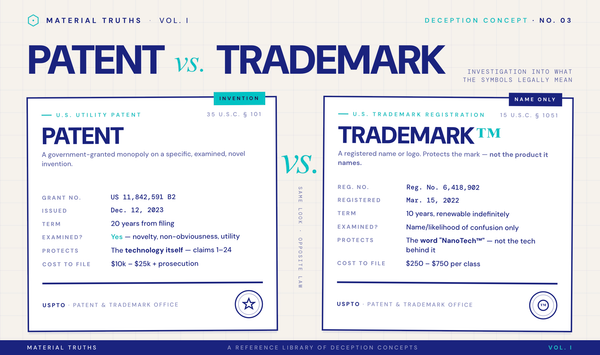
Is proprietary blend the same as trademarked? No. Proprietary blend refers to the labeling format; a trademark protects the blend's name only.
Can a proprietary blend be patented? Theoretically yes, but most are not. Verify any patent claim at USPTO.gov.
How do I know what's really in a proprietary blend? Usually you cannot, without manufacturer disclosure or independent testing. Third-party certifications (NSF, USP, Informed Sport) confirm label accuracy but not necessarily individual component amounts.
Further reading
- Sciencewashing: The broader pattern proprietary blends fit into
- Patent vs. Trademark: Why "trademarked blend" is not "patented blend"
- Claims decoder: "Proprietary": How "proprietary" is misused across consumer products
- Claims decoder: "Clinical-grade": The adjacent language trap
Sources
- Dietary Supplement Health and Education Act of 1994 (DSHEA), Public Law 103-417. fda.gov/food/dietary-supplements/dietary-supplement-health-and-education-act-1994
- FDA. 21 CFR 101.36: Nutrition labeling of dietary supplements. ecfr.gov
- FDA. "Current Good Manufacturing Practice in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements." 21 CFR Part 111.
- NSF International. "Certified for Sport." nsfsport.com
- U.S. Pharmacopeia. "Dietary Supplement Verification Program." usp.org
Get notified when we add new enforcement examples to this page.