Sciencewashing: What It Is, How to Spot It, and Why It Works

Last updated: April 20, 2026
Sciencewashing is the use of clinical-sounding language, technical terminology, or cherry-picked data to make ordinary products appear scientifically advanced when they are not. It typically involves inventing proprietary names for commodity materials, citing internal studies as independent research, and invoking scientific concepts that sound authoritative but mean little. Consumers encounter it most often in supplements, skincare, bedding, and wellness products.
What is sciencewashing?
Sciencewashing is a marketing strategy that exploits a specific asymmetry: most consumers cannot distinguish legitimate scientific language from fabricated scientific language in the time it takes to make a purchase. A product labeled with phrases like "clinical-grade formula," "proprietary Nano-matrix technology," or "dermatologist-validated active ingredients" is functionally indistinguishable from one that actually has scientific substantiation — unless the consumer investigates the primary sources.
The term "sciencewashing" parallels "greenwashing" (deceptive environmental claims) and "pinkwashing" (deceptive breast cancer awareness marketing). All three describe the practice of using surface-level credibility signals to mask underlying claims that would not survive scrutiny.
Sciencewashing is distinct from outright fraud. A product can be sciencewashed while containing no outright lies — the deception operates at the level of implication, emphasis, and selective disclosure. The Federal Trade Commission evaluates deceptive advertising based on the "overall net impression" conveyed to a reasonable consumer, not on any single literal statement.
How does sciencewashing typically work?
Five distinct tactics appear repeatedly across industries.
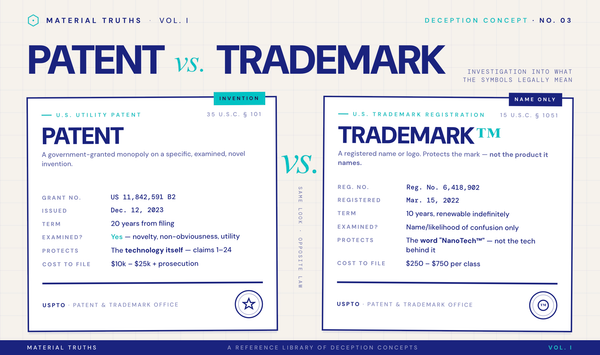
1. Trademarked names for commodity materials. A brand trademarks a name like "ThermaTech™" or "NanoActive®" to describe what is, on examination, ordinary polyester, bamboo viscose, or another commodity ingredient. The ™ and ® symbols cost a few hundred dollars to obtain and protect only the brand name, not any underlying technology. Consumers often interpret these symbols as signals of patented innovation. As we cover in our reference page on patent vs. trademark, the distinction is legally precise but marketed ambiguously.
2. "Clinically studied" instead of "clinically proven." A brand can truthfully claim a product was "clinically studied" even when the study found no benefit, had methodological weaknesses, or was never peer-reviewed. "Proven" implies demonstrated efficacy; "studied" implies only inclusion in some testing. Consumers consistently misremember "studied" as "proven," and brands know this. See our page on clinically tested vs. clinically proven for the full breakdown.
3. Internal testing presented as independent research. Brands commission their own studies, frame the results as "research-backed," and omit that the research was conducted in-house or by a paid contract lab. Legitimate independent research is conducted by universities, government agencies, or third-party organizations with no financial stake in the outcome. Brand-funded research can still be valid, but it carries a different evidentiary weight and should be disclosed as such.
4. Invocation of scientific institutions without affiliation. Phrases like "developed with NASA technology," "formulated by Harvard researchers," or "based on MIT studies" often refer to tenuous connections — a single consulting engagement, a researcher who once attended the institution, or research that was simply published by someone affiliated with that institution but has no endorsement relationship. Institutions almost never endorse consumer products; when they do, the endorsement is documented and verifiable.
5. Technical jargon obscuring commodity formulation. A skincare product might describe its "tri-peptide micro-delivery system" to describe what is, in plain language, a moisturizer with three peptides dissolved in it. The jargon is technically accurate but designed to obscure that the underlying formulation is neither novel nor distinctive.
What are real examples of sciencewashing in consumer products?
Editorial note: This section will be populated with links to Material Truths investigations as they publish. Initial examples below are drawn from publicly documented enforcement actions.
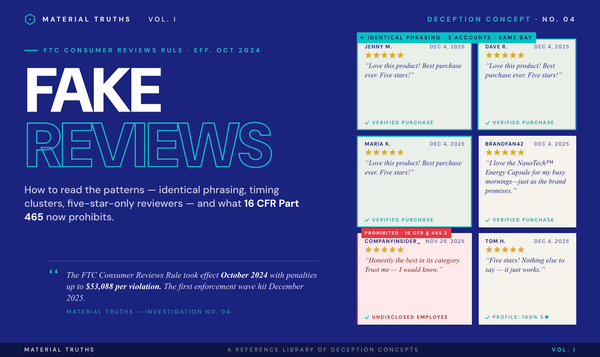
The Federal Trade Commission has brought numerous cases where product claims crossed from sciencewashing into actionable deception. Documented patterns include supplement brands citing studies that were never published, technology claims that could not be substantiated when challenged, and "clinical-grade" claims attached to products that had undergone no clinical evaluation.
Material Truths investigations into brands such as Rest Bedding, Nuzzle Pillow, and Mellow Sleep have documented specific examples of trademarked names presented as patented technology, internal tests cited as "research," and fabricated institutional connections. Each investigation includes the primary-source evidence — USPTO records, corporate filings, and direct comparisons between marketing language and documented facts.
How can consumers spot sciencewashing?
A practical checklist:
- If a product claims a "proprietary" technology, ask: is there a patent number? A real patent is findable in the USPTO Trademark and Patent database. If the brand cannot produce a patent number, the "technology" is almost certainly a trademarked name for a commodity material.
- If a product is "clinically studied," ask: what did the study conclude? Legitimate studies have publicly available abstracts, typically on PubMed or in the journal that published them. A brand that cannot provide a study citation is relying on the ambiguity of "studied."
- If a product references a scientific institution, ask: is the affiliation verifiable? University technology transfer offices publish lists of licensed brands. If a product claims "NASA technology," NASA's technology transfer program has a searchable database.
- Look for weasel words. Phrases like "helps support," "may promote," "designed to assist," and "engineered to enhance" are legally protective because they make no specific factual claim. They are not evidence of efficacy; they are evidence of claims calibrated to avoid substantiation.
- Check who funded any cited research. Published studies include funding disclosures. Brand-funded studies are not invalid, but they should be weighed differently than independent research.
- Search the brand's name plus "FTC" or "NAD." Prior enforcement actions signal a pattern. The FTC maintains a searchable public database of consumer protection cases at ftc.gov.
What does the law say about sciencewashing?
The primary federal authority is Section 5 of the Federal Trade Commission Act, which prohibits "unfair or deceptive acts or practices in or affecting commerce." Under Section 5, advertisers must have a reasonable basis for objective claims before they are made — a requirement known as the "prior substantiation" doctrine. For health, efficacy, or technology claims, the FTC typically requires "competent and reliable scientific evidence," defined as tests, analyses, research, or studies conducted by qualified persons using procedures generally accepted in the relevant field.
The FTC evaluates advertising based on the "overall net impression" a reasonable consumer would take from the advertisement as a whole, not on any single literal statement. This means a technically true claim can still be deceptive if the overall impression is misleading — a central principle in how sciencewashing cases are built.
The National Advertising Division (NAD) of BBB National Programs provides a self-regulatory forum where competitors can challenge one another's advertising claims. NAD proceedings are faster than FTC enforcement and produce published decisions that become precedent. Many sciencewashing cases are resolved at NAD before ever reaching the FTC.
State consumer protection laws — California's Consumers Legal Remedies Act, New York's General Business Law §349, and similar statutes in most states — provide additional enforcement pathways, often through class action litigation.
Recent enforcement actions
The FTC's 2023 overhaul of its Endorsement Guides tightened requirements around testimonials and third-party scientific claims. Subsequent enforcement has focused on supplement brands making unsubstantiated health claims, consumer technology brands claiming patents they do not hold, and beauty brands invoking "clinical" testing that did not exist.
Class actions have also emerged as a significant enforcement vector. Product categories where sciencewashing claims have been successfully challenged in court include cooling bedding, wearable wellness devices, "detoxifying" skincare, and performance supplements.
This section is updated quarterly as new enforcement actions are documented.
What's the difference between sciencewashing and greenwashing?
The two practices are mechanically similar but operate in different domains:
| Dimension | Sciencewashing | Greenwashing |
|---|---|---|
| Core claim type | Technology, efficacy, clinical | Environmental, sustainability |
| Primary regulator | FTC (Section 5, prior substantiation) | FTC (Green Guides), state AGs |
| Typical signals | Trademarked "technologies," clinical language | "Eco-friendly," "natural," "recyclable" |
| Detection method | USPTO search, study verification | Certification verification, supply chain docs |
| Industries most affected | Supplements, skincare, bedding, electronics | Food, household products, apparel |
A single product can be both sciencewashed and greenwashed simultaneously. A "clinically tested, eco-friendly antibacterial blanket" using an unpatented material with no EPA registration and no independent environmental certification would trigger both deception patterns.
Frequently asked questions
What is sciencewashing? Sciencewashing is the use of clinical-sounding language, technical terminology, or cherry-picked data to make ordinary products appear scientifically advanced when they are not. See the "What is sciencewashing?" section above for full detail.
Is sciencewashing the same as greenwashing? No. Sciencewashing covers misleading scientific, technological, and clinical claims broadly; greenwashing specifically covers misleading environmental claims. See the comparison table above.
Is sciencewashing illegal? Sciencewashing becomes actionable under Section 5 of the FTC Act when the claims are false or unsubstantiated. Not every instance of sciencewashing rises to the level of illegal deception, but the dividing line is thinner than most brands assume.
What does "clinically studied" mean? It means a product was included in some form of clinical testing. It does not mean the product was shown to work.
How can I verify scientific claims on a product? Ask for the published study, search PubMed for the citation, verify any patent claims at USPTO.gov, and check whether cited scientific institutions actually endorse the product.
Can a product be sciencewashed and still work? Yes. Sciencewashing describes misleading marketing, not necessarily product ineffectiveness. A product may actually work while being marketed deceptively.
Further reading
- Proprietary Blend: The specific sciencewashing pattern used in supplements
- Patent vs. Trademark: Why the distinction matters for technology claims
- Clinically Tested vs. Clinically Proven: The specific language trap
- Claims decoder: "Patented": How to verify any patent claim
Sources
- Federal Trade Commission. "Advertising FAQ's: A Guide for Small Business." ftc.gov/business-guidance/resources/advertising-faqs-guide-small-business
- Federal Trade Commission. "Guides Concerning the Use of Endorsements and Testimonials in Advertising" (updated 2023). 16 CFR Part 255.
- BBB National Programs, National Advertising Division. Decisions database. bbbprograms.org/programs/all-programs/national-advertising-division
- USPTO Trademark Status & Document Retrieval. tsdr.uspto.gov
Get notified when we add new enforcement examples to this page. Material Truths sends a monthly update when new FTC, NAD, or class action rulings are added to our reference pages.