What "Antimicrobial" Legally Means on Consumer Products (and What It Doesn't)

Last updated: May 7, 2026
"Antimicrobial" is one of the most heavily regulated marketing claims in U.S. consumer products. The EPA treats any substance intended to kill or mitigate microorganisms as a pesticide under the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA). Products making public health claims about protecting users from germs must be registered with EPA and submit efficacy data. Products using the Treated Articles Exemption can only claim to protect the article itself — not the user. Most "antimicrobial" consumer products on the market today are using one of these two regulatory pathways, and which pathway determines what the brand can legally say.
What does "antimicrobial" legally mean?
Under FIFRA Section 2(mm), an "antimicrobial pesticide" is any substance intended to disinfect, sanitize, reduce, or mitigate the growth of microorganisms. This includes bacteria, viruses, fungi, mold, and mildew. Because the law treats microorganisms as "pests," any product designed to act against them falls under pesticide regulation.
EPA divides antimicrobial products into two categories based on the claims made:
Public health antimicrobials. Products making claims about protecting people from disease-causing microorganisms — "kills 99.9% of germs," "reduces risk of illness," "protects against pathogens." These require EPA registration with submitted efficacy data demonstrating the claim is scientifically supported.
Non-public health antimicrobials. Products making claims about economic or aesthetic effects — preventing fabric degradation, stopping mold on paint, inhibiting odor-causing bacteria on a product's surface. These still require EPA registration of the pesticide ingredient, but the required data is less extensive.
A finished consumer product (a towel, a shower curtain, a cutting board) that contains an antimicrobial can fall under either category depending on the claims made about it. The same underlying chemistry can be marketed legally or illegally based purely on what the brand says about it.
Who regulates antimicrobial claims?
Jurisdiction depends on the product type:
- EPA (primary for most consumer products). FIFRA governs claims about protecting people, animals, or property from microorganisms. Enforcement has intensified since the COVID-19 pandemic significantly increased consumer-facing antimicrobial marketing.
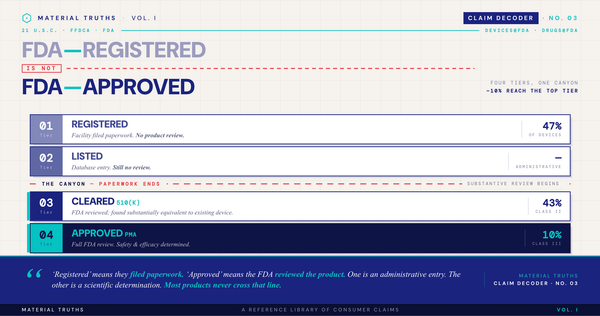
- FDA (medical devices). Surgical gowns, wound dressings, implants, and other medical devices with antimicrobial features require FDA 510(k) premarket clearance.
- FDA (certain cosmetics and drugs). Antimicrobial claims on drug products (hand sanitizers, antibacterial soaps) fall under FDA drug regulation.
- State agencies. California's Department of Pesticide Regulation requires separate state registration for antimicrobial products sold in the state. Other states have varying requirements.
- FTC. Concurrent authority to pursue unsubstantiated marketing claims under Section 5 of the FTC Act, even when EPA registration is not at issue.
The multi-agency framework means a brand can be compliant with one regulator while violating another's rules.
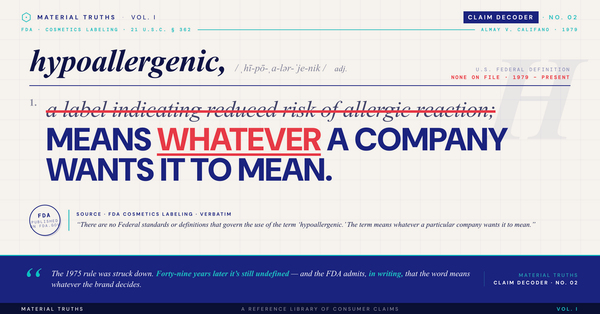
What's the difference between "antimicrobial" and "antibacterial"?
The terms are not interchangeable in regulatory context:
| Term | What it covers |
|---|---|
| Antimicrobial | Broad — bacteria, viruses, fungi, mold |
| Antibacterial | Bacteria only |
| Antiviral | Viruses only |
| Antifungal | Fungi only |
| Antimycobacterial | Mycobacteria (e.g., tuberculosis) — specific registration |
A product registered with EPA for antibacterial efficacy cannot claim antiviral properties without submitting separate efficacy data for viruses. Marketing that conflates the terms — using "antimicrobial" to imply both antibacterial and antiviral protection when only antibacterial data was submitted — violates FIFRA.
What is the Treated Articles Exemption?
Many consumer products claim antimicrobial properties through the Treated Articles Exemption at 40 CFR 152.25(a), clarified by EPA Pesticide Registration Notice 2000-1. The exemption allows products to use an EPA-registered antimicrobial without separately registering the finished product — but only under specific conditions:
- The antimicrobial ingredient itself must be EPA-registered for the specific use
- The treatment must be intended to protect the article itself, not the user
- No public health claims can be made about the finished product
Acceptable language under the exemption, per EPA guidance:
"Antimicrobial properties are built in to inhibit the growth of bacteria that may affect this product. The antimicrobial properties do not protect users or others against bacteria, viruses, germs, or other disease organisms."
Explicitly prohibited language includes "fights germs," "kills 99.9%," "self-sanitizing," "self-disinfecting," "hospital-grade," and any implication that the product protects people from illness. EPA enforcement specifically targets this type of claim inflation.
How can consumers verify an antimicrobial claim?
1. Check for an EPA registration number. Products making public health antimicrobial claims must display an EPA Registration Number ("EPA Reg. No.") on the label. Format: XXXXX-XXX or XXXXX-XXX-XXXXX.
2. Search the EPA Pesticide Product Label System (PPLS). Located at iaspub.epa.gov/apex/pesticides/f?p=PPLS:1. Enter the EPA Reg. No. or the company name. Registered products appear with their approved labels, including the specific claims EPA has authorized.
3. Ask the brand for the EPA Reg. No. of the treatment. For treated articles (products using the Treated Articles Exemption), the antimicrobial treatment itself must be EPA-registered. A legitimate brand can provide the registration number of the treatment ingredient (for example, Silpure, Microban, Sanitized, Polygiene, etc., each have EPA registrations for specific uses).
4. Verify state registration if applicable. California's Department of Pesticide Regulation maintains a product database at cdpr.ca.gov. Products sold in California making antimicrobial claims should have separate state registration.
5. Compare marketing claims to approved label language. If EPA has approved the treatment for odor control only but the brand markets it as "kills germs," the marketing exceeds the registration and is a FIFRA violation.
Common antimicrobial claims that cross the line
Examples of claims that typically indicate non-compliance:
- "Kills 99.9% of bacteria" — public health claim requiring registration
- "Self-sanitizing fabric" — self-sanitizing implies sanitizer efficacy, requires registration
- "Hospital-grade antimicrobial" — medical/public health claim requiring registration
- "Protects your family from germs" — public health claim requiring registration
- "Inhibits the growth of dangerous bacteria" — "dangerous" implies public health
- "Antiviral protection" — requires separate antiviral registration
Claims that may be compliant under the Treated Articles Exemption:
- "Antimicrobial properties inhibit the growth of odor-causing bacteria"
- "Built-in freshness protection prevents fabric deterioration"
- "Contains an antimicrobial preservative to protect the product"
Recent enforcement on antimicrobial claims
EPA has significantly increased enforcement since the COVID-19 pandemic. Notable patterns:
- Textile and apparel products claiming to protect users from viruses or bacteria have received warning letters and stop-sale orders
- E-commerce platform listings with "antiviral" or "self-sanitizing" claims have been removed following EPA complaints
- Individual distributors have been held liable even when they did not manufacture the treated product — FIFRA places liability throughout the distribution chain
- EPA issued new guidance in August 2023 on soft surface textiles in non-residential settings, creating a formal framework for clinical and institutional antimicrobial textile registrations
This section is updated as new enforcement actions are documented.
Further reading
- Sciencewashing: Antimicrobial marketing often overlaps
- Claim decoder: "Clinical-Grade": Adjacent term
- Antimicrobial claim decoder: Where "hospital-grade" has specific regulatory meaning
Sources
- EPA. "Consumer Products Treated with Pesticides." epa.gov/safepestcontrol/consumer-products-treated-pesticides
- 40 CFR 152.25(a) — Treated Articles Exemption.
- EPA Pesticide Registration Notice 2000-1. epa.gov/sites/default/files/2014-04/documents/pr2000-1.pdf
- EPA. "Pesticide Registration Manual: Chapter 4 - Antimicrobial Products." epa.gov/pesticide-registration
- EPA Pesticide Product Label System. iaspub.epa.gov/apex/pesticides/f?p=PPLS:1
- AATCC Test Method 100, ISO 20743, ASTM E2149 — antimicrobial textile testing standards.