FDA-Registered vs. FDA-Approved: The Difference That Decides If a Claim Is Real
Last updated: May 8, 2026
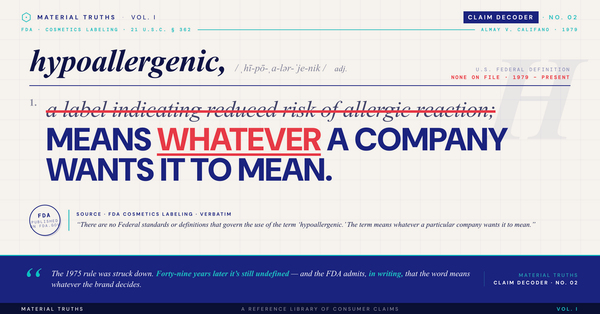
"FDA-registered" means a company filed paperwork with the FDA. "FDA-approved" means the FDA reviewed the product and determined it safe and effective. The two claims communicate vastly different things — and the FDA itself has publicly called the confusion misleading. Only about 10 percent of medical devices are "FDA-approved." Most cosmetics, dietary supplements, and consumer wellness products are not approved by the FDA at any level, even when marketing suggests otherwise. This decoder walks through the four regulatory tiers, how to verify a claim in 30 seconds, and what the FDA does not approve at all.
What does "FDA-registered" actually mean?
Under the Federal Food, Drug, and Cosmetic Act, domestic and foreign facilities that produce food, drugs, medical devices, and certain other FDA-regulated products must register with the FDA annually. This registration is an administrative requirement — the FDA needs to know what facilities exist in the supply chain so it can inspect them and track products during recalls.
The FDA's official position, published on its own website at fda.gov, is unambiguous: "When a facility registers its establishment and lists its devices, the resulting entry in the FDA's registration and listing database does not denote approval, clearance, or authorization of that facility or its medical devices."
The agency has gone further. On March 3, 2021, the FDA issued a formal notice titled "FDA Calls on Certain Firms to Stop Producing and Issuing Misleading 'FDA Registration Certificates.'" The FDA explicitly stated that it does not issue registration certificates, and that firms displaying such certificates alongside product marketing to imply FDA review misbrand their products in violation of federal law.
A related technical point the FDA has flagged: devices get "listed" while establishments get "registered." Marketing a specific product as "FDA-registered" is not just misleading — it's technically incorrect, and falls under misbranding provisions at 21 CFR 207.77.
The four FDA status tiers
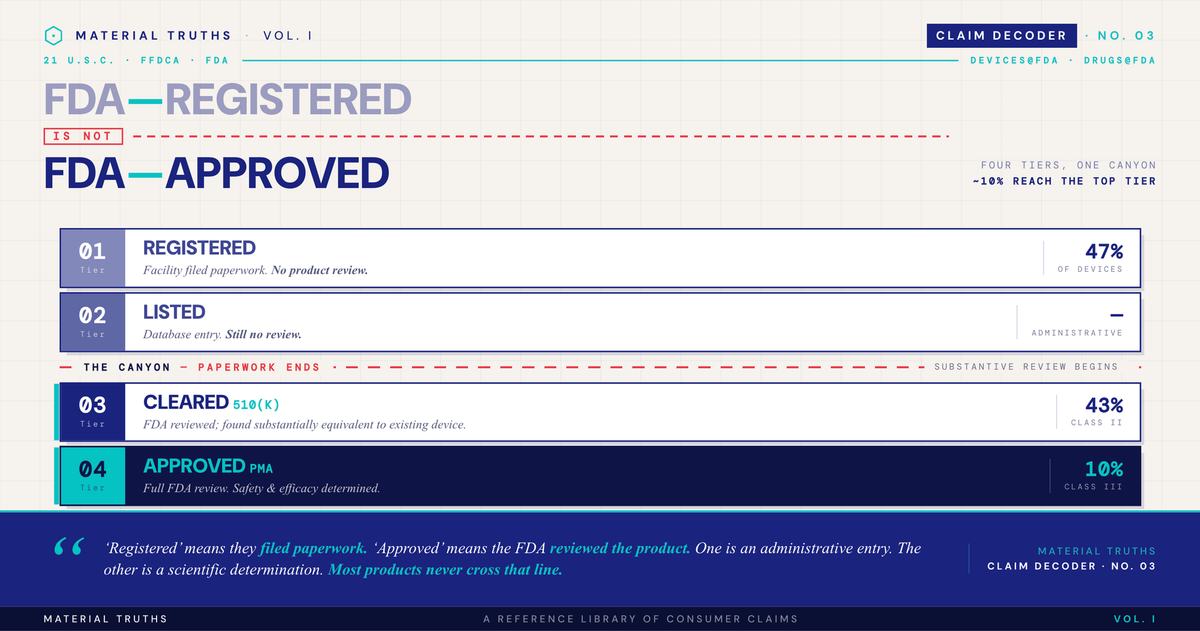
FDA medical device regulation involves four progressively rigorous tiers:
| Tier | What it requires | Approximate share of devices | Consumer meaning |
|---|---|---|---|
| FDA-registered / FDA-listed | Administrative filing only | 47% (Class I) | The FDA knows the facility exists |
| FDA-cleared (510(k)) | "Substantially equivalent" to existing device | 43% (Class II) | Compared to a predicate device; minimal independent testing |
| FDA-granted (De Novo) | New low/moderate-risk device, no predicate | Small fraction | Novel pathway for devices without comparators |
| FDA-approved (PMA) | Rigorous FDA review of safety and effectiveness | 10% (Class III) | FDA affirmatively determined safe and effective |
Only "cleared" and "approved" involve substantive FDA review. "Registered" and "listed" are essentially the regulatory equivalent of filing paperwork to operate a business.
The terminology matters. A Class II device is "FDA-cleared," not "FDA-approved." Marketing a cleared device as "approved" is false advertising under the Lanham Act (15 U.S.C. § 1125(a)) and state consumer protection laws. Courts have consistently upheld this distinction as material — the approved/cleared difference affects purchase decisions for health-related products.
What FDA does not approve at all
Large categories of consumer products are not subject to FDA pre-market approval, despite consumer expectations:
1. Dietary supplements. Under the Dietary Supplement Health and Education Act (DSHEA) of 1994, supplements do not require FDA approval before going to market. Manufacturers must register their facilities, comply with manufacturing standards, and avoid specific prohibited ingredients, but FDA does not review efficacy or safety before supplements are sold. The FDA itself states: "If a supplement promises a cure or quick fix for a health problem, it may be too good to be true."
2. Cosmetics. With the narrow exception of color additives, FDA does not approve cosmetics before market. Ingredient restrictions and labeling requirements apply, but there is no product review process. Claims that cosmetics are "FDA-approved" are essentially always false.
3. Most foods. FDA does not approve food products or food labels before they reach market. Manufacturers must comply with labeling and safety regulations, but there is no per-product approval. Food additives that have not achieved GRAS (Generally Recognized As Safe) status do require approval, but the food product itself generally does not.
4. Over-the-counter drugs meeting OTC monograph standards. OTC drugs are evaluated against pre-established active ingredient monographs and do not undergo individual product review. They're technically compliant with FDA standards rather than approved.
5. Cosmetic-adjacent products. Soaps, many personal care items, and products marketed for non-medical purposes fall outside FDA pre-market review.
Because these categories dominate consumer retail — bedding, skincare, wellness, personal care — most products sold directly to consumers have not been FDA-approved and should not make such claims.
How to verify an FDA claim in 30 seconds
For medical devices: Search the Devices@FDA database at accessdata.fda.gov/scripts/cdrh/devicesatfda/. Enter the brand name or device name. Legitimate cleared or approved devices appear with a specific 510(k) number (starts with "K") or PMA number (starts with "P"). If the device doesn't appear in this database, the FDA has not cleared or approved it.
For drugs: Search Drugs@FDA at accessdata.fda.gov/scripts/cder/daf/. Legitimate FDA-approved drugs appear with NDA (New Drug Application) or ANDA (Abbreviated New Drug Application) numbers.
For establishment registration (facility filing only): Search FDA's establishment registration and device listing database at accessdata.fda.gov/scripts/cdrh/cfdocs/cfRL/rl.cfm. A facility appearing here has registered — that's all it means. The facility registration does not extend to the products.
Warning signs of fake or misleading claims:
- A "registration certificate" displayed on a product page, store, or marketing material. FDA does not issue such certificates.
- Use of the FDA logo. The logo is reserved for official government use; private use to suggest endorsement is prohibited.
- "FDA-approved" claims on cosmetics, dietary supplements, or food products (with very narrow exceptions).
- "FDA-registered device" used as if it were meaningful. Devices are "listed" not "registered," and listing alone is not review.
- References to an NDC (National Drug Code) number presented as evidence of approval. NDC numbers are administrative identifiers and do not indicate approval.
Recent enforcement on false FDA claims
Enforcement has been steady and multi-vector:
- FDA warning letters. FDA routinely sends warning letters for false FDA-approved or FDA-registered claims, particularly for dietary supplements making drug-level claims and for medical device facilities issuing fake registration certificates.
- Lanham Act competitor suits. Competitors regularly sue for false FDA-approved claims, because the approved/cleared distinction is material to purchase decisions. Courts have repeatedly sustained such claims.
- State consumer protection class actions. California's Consumers Legal Remedies Act, New York General Business Law §§ 349 and 350, and similar statutes have produced significant class action settlements against brands making false FDA claims.
- FTC enforcement. When FDA claims are part of broader deceptive advertising, the FTC can pursue under Section 5 of the FTC Act.
This section is updated as new enforcement actions are documented.
Frequently asked questions
What does "FDA-registered" actually mean? A facility filed registration paperwork. The FDA explicitly states this does not indicate approval, clearance, or authorization of products.
Difference between FDA-registered, FDA-cleared, and FDA-approved? Registered = paperwork; cleared = substantially equivalent to existing device (Class II); approved = rigorous review (Class III only).
Do dietary supplements need FDA approval? No. Supplements do not require pre-market approval under DSHEA 1994.
Does FDA approve cosmetics? No, with narrow exceptions for color additives.
How can I verify an FDA claim? Search Devices@FDA for devices or Drugs@FDA for drugs. If the product isn't there, FDA hasn't approved or cleared it.
Has FDA taken action against false claims? Yes, including a March 2021 statement specifically on misleading FDA registration certificates.
Further reading
- Sciencewashing: Broader pattern where FDA-registration claims appear
- Claim decoder: "Patented": Adjacent verification-dependent claim
- Claim decoder: "Clinical-Grade": Related medical-adjacent term
- Clinically Tested vs. Clinically Proven: Related substantiation language
Sources
- FDA. "Are There 'FDA Registered' or 'FDA Certified' Medical Devices? How Do I Know What Is FDA Approved?" fda.gov/medical-devices/consumers-medical-devices
- FDA. "FDA Calls on Certain Firms to Stop Producing and Issuing Misleading 'FDA Registration Certificates.'" March 3, 2021.
- FDA. "Is It Really 'FDA Approved'?" fda.gov/consumers/consumer-updates/it-really-fda-approved
- FDA. "10 Facts about What FDA Does and Does Not Approve." fda.gov/consumers/consumer-updates
- Devices@FDA. accessdata.fda.gov/scripts/cdrh/devicesatfda/
- Drugs@FDA. accessdata.fda.gov/scripts/cder/daf/
- 21 CFR 207.77 — Misbranding provisions.
- Lanham Act, 15 U.S.C. § 1125(a).