What "Hypoallergenic" Legally Means (Hint: Almost Nothing)
Last updated: May 7, 2026
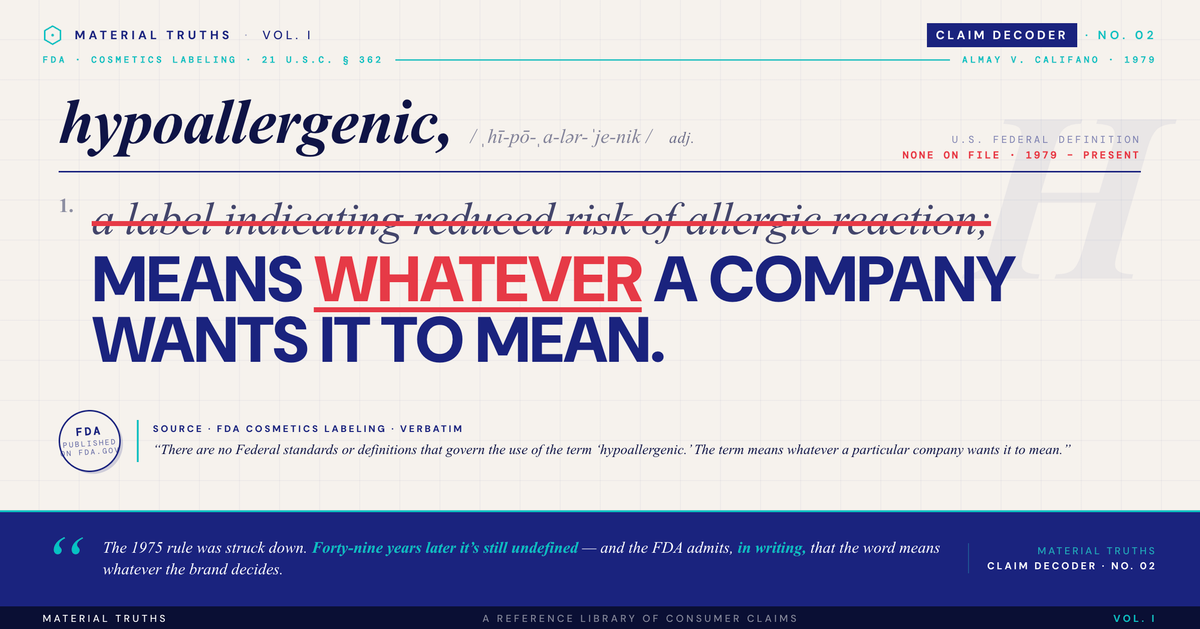
"Hypoallergenic" has no legal definition in U.S. cosmetic regulation. The FDA explicitly acknowledges on its consumer information pages that the term "means whatever a particular company wants it to mean." There are no testing requirements, no government certification, and no regulatory verification. A brand can apply the label to any product — cosmetics, bedding, pet products, baby products, cleaning supplies — without demonstrating any reduction in allergic reaction risk. The gap between what the word appears to promise and what it legally requires is one of the largest in consumer product marketing.
What does "hypoallergenic" legally mean?
Nothing. Not as a regulatory matter.
The Federal Food, Drug, and Cosmetic Act governs cosmetic labeling through 21 U.S.C. § 362. The FDA has authority to prohibit "false or misleading" labeling. But the FDA has no specific regulation defining "hypoallergenic" or requiring any testing before a product can use the term.
This means:
- A cosmetic manufacturer can label any product "hypoallergenic" without conducting any allergy testing
- No government agency verifies the claim
- No minimum ingredient restrictions apply to products using the term
- Ingredients known to cause allergic reactions can remain in products labeled hypoallergenic
- The only limit is the FTC's general prohibition on false or misleading advertising — which requires proving a specific consumer was misled in a material way
In practice, "hypoallergenic" functions as a marketing term with the regulatory weight of "delicious" or "refreshing." It signals intent, not verified fact.
Did the FDA ever try to regulate "hypoallergenic"?
Yes. In 1975, the FDA proposed regulations at 21 CFR 701.20 that would have required cosmetics labeled "hypoallergenic" to undergo testing demonstrating a lower incidence of adverse reactions compared to conventional products. The rule was an attempt to give the term specific regulatory meaning.
Almay and Clinique challenged the regulations in court. In Almay Inc. v. Califano, 613 F.2d 1070 (D.C. Cir. 1979), the D.C. Circuit Court of Appeals struck down the regulations. The court held that the FDA had not adequately supported its interpretation of what "hypoallergenic" communicated to consumers or demonstrated the regulatory framework's necessity.
The FDA never reissued the rule. Nearly fifty years later, "hypoallergenic" remains legally undefined. The FDA's current Cosmetic Product Labeling page on fda.gov specifically addresses this: "There are no Federal standards or definitions that govern the use of the term 'hypoallergenic.' The term means whatever a particular company wants it to mean."
Does "hypoallergenic" mean the product won't cause allergic reactions?
No.
Even when brands voluntarily conduct testing to support their hypoallergenic claims, the testing has significant limits:
1. Testing on small populations. Typical hypoallergenic testing involves 50 to 200 participants in a Repeated Insult Patch Test (RIPT) or similar protocol. The absence of reactions in this small sample does not predict the absence of reactions in the broader population.
2. Controlled conditions. Patch tests apply products to the skin under controlled conditions that may not replicate real-world use. A product that doesn't irritate in a patch test may still cause reactions during actual prolonged use.
3. Individual variation. Allergic responses are highly individual. A product tolerated by 99 percent of tested users can still cause severe reactions in susceptible individuals. No test design can rule out all possible allergic reactions.
4. Specific allergens not always addressed. General testing doesn't verify absence of specific allergens like fragrance compounds, preservatives, or specific proteins. A person with a known allergen should review ingredient lists, not rely on the hypoallergenic label.
What should consumers look for instead?
More meaningful signals than the unregulated "hypoallergenic" label:
1. Ingredient list review. U.S. cosmetic regulations do require ingredient labeling (with limited exceptions for professional-use products). Review the INCI ingredient list for known allergens relevant to your situation: fragrance/parfum, parabens, formaldehyde-releasing preservatives (DMDM hydantoin, quaternium-15), lanolin, specific botanicals. For textiles, review fiber content and any treatments disclosed.
2. Legitimate certifications that actually require testing:
- National Eczema Association Seal of Acceptance (nationaleczema.org/eczema-products) — voluntary program that evaluates products for irritation potential in eczema-prone skin using defined criteria.
- OEKO-TEX STANDARD 100 (oeko-tex.com) — independent textile certification testing for 100+ harmful substances.
- USDA Organic — verified absence of synthetic pesticides and prohibited substances in food and agricultural products.
3. Professional testing services. Some dermatology practices offer product patch testing to identify specific sensitivities before regular use.
4. Small-area testing. Before using any new product on broader skin or as bedding, test on a small area for 24 to 48 hours.
5. Patient-specific allergen lists. Resources like the American Contact Dermatitis Society (contactderm.org) maintain databases of common allergens and products that avoid them.
Do other sensitivity claims have legal meaning?
No. Similar terms with no regulatory definition:
- "Dermatologist tested" — means a dermatologist tested the product. Nothing about outcome.
- "Dermatologist recommended" — means at least one dermatologist recommends it.
- "Sensitive skin formula" — marketing; no specific formulation requirements.
- "Allergy tested" — means some form of testing occurred. Outcome unspecified.
- "Non-irritating" — no regulatory standard for irritation testing.
- "Gentle" — purely promotional.
- "For sensitive skin" — no specific requirements.
All of these are legally equivalent to "hypoallergenic" in terms of regulatory rigor: they sound reassuring but require nothing specific from the manufacturer.
Recent enforcement on hypoallergenic and related claims
Because "hypoallergenic" has no legal standard, direct FTC enforcement is rare. Enforcement patterns:
- FTC false advertising actions. When brands combine "hypoallergenic" with specific substantiation-based claims (e.g., "clinically proven hypoallergenic," "99% of sensitive skin users reported no irritation"), the specific substantive claims can be challenged under Section 5 even if "hypoallergenic" alone cannot.
- State consumer protection actions. California's Consumers Legal Remedies Act, New York General Business Law §349, and similar statutes have supported class actions where hypoallergenic claims accompanied specific factual assertions.
- FDA warning letters. When hypoallergenic claims cross into drug claims (implying treatment of allergic conditions), FDA can pursue the drug claim under the Food, Drug, and Cosmetic Act.
The gap between the term's regulatory weight and consumer interpretation remains. Legislative proposals to define the term have been introduced in Congress over the years but have not advanced. The FDA has not reopened the rulemaking process since the Almay decision.
Frequently asked questions
What does "hypoallergenic" legally mean? Nothing specific. The FDA acknowledges the term has no Federal standard.
Did the FDA ever try to regulate it? Yes, in 1975 (21 CFR 701.20), but the regulations were struck down in Almay v. Califano (1979) and never reinstated.
Does it mean the product won't cause allergic reactions? No. Any product can cause an allergic reaction in a susceptible person.
Are there legitimate certifications? Not for "hypoallergenic" itself. But the National Eczema Association Seal of Acceptance, OEKO-TEX STANDARD 100, and USDA Organic do involve real testing.
What should I look for instead? Direct ingredient review, meaningful third-party certifications, and professional patch testing for known sensitivities.
Do "dermatologist tested" or "sensitive skin" have legal meaning? No — all similar sensitivity claims lack specific regulatory definition.
Further reading
- Sciencewashing: Broader pattern of pseudoscientific marketing
- Clinically Tested vs. Clinically Proven: Related language trap
- Claim decoder: "Natural Fragrance": Adjacent unregulated term
- Claim decoder: "Dermatologist-Tested": Adjacent unregulated term
Sources
- FDA. "'Hypoallergenic' Cosmetics." fda.gov/cosmetics/cosmetics-labeling-claims/hypoallergenic-cosmetics
- FDA. "Cosmetic Labeling Claims." fda.gov/cosmetics/cosmetics-labeling-claims
- Almay Inc. v. Califano, 613 F.2d 1070 (D.C. Cir. 1979).
- Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 362.
- National Eczema Association. "Seal of Acceptance Program." nationaleczema.org/eczema-products
- OEKO-TEX. oeko-tex.com